|
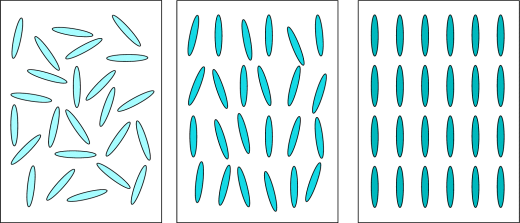
In crystalline solids the position and orientation of atoms and molecules are relative to each other. They are rigidly held in position by strong intermolecular or ionic forces and chemical bonding in fixed geometric patterns or lattices.
The molecules within a liquid are moving constantly within the volume, exhibiting a large degree of disorder and rapid fluctuations of position and orientation.
In liquid crystals the molecules display various degrees of order between that of isotropic liquid and crystalline solid. The various liquid crystal phases can be characterized by the type of ordering that is present: positional order where molecules are arranged in any kind of ordered lattice and orientational order where molecules are mostly pointing in the same direction. The order can be either short-range, only between molecules close to each other, or long-range which extends to larger, sometimes macroscopic dimensions.
Liquid crystals can be divided into thermotropic and lyotropic. Thermotropic liquid crystals exhibit a phase transition into the liquid crystals phase as temperature is changed, whereas lyotropic liquid crystals exhibit phase transitions as a function of the concentration of micelles as well as temperature.
The amphiphilic molecules containing, hydrophilic and hydrophobic parts within the same molecule, assemble into micelles. These spherical objects do not order themselves in solution. However, at higher concentration, the assemblies will become ordered.
In contrast to thermotropic liquid crystals, lyotropic liquid crystals have another degree of freedom of concentration that enables them to induce a variety of different phases.
- Discontinuous cubic phase (micellar phase)
- Hexagonal columnar phase (middle phase)
- Bicontinuous cubic phase
- Lamellar phase
- Bicontinuous cubic phase
- Reverse hexagonal columnar phase
- Inverse cubic phase (Inverse micellar phase)
|